Suki W, Zabaneh R, Cangiano J, Reed J, Fischer D, Garrett L, Ling B et al. Effects of sevelamer and calcium-based phosphate binders on mortality in hemodialysis patients. Kidney Int; 29 August 2007; doi:10.1038/sj.ki.5002466
Effects of sevelamer and calcium-based phosphate binders on mortality in hemodialysis patients
Background
DCOR was an effectiveness trial that was designed to evaluate the treatment outcomes under usual care conditions in the hemodialysis population.
Trial Design
It was a multicenter, randomized, open-label effectiveness trial stratified by race (black and non-black), sex, diabetic status, and by age (<55 and >=55).
Population
This study included 2013 patients who were were >18 years, on dialysis for >3 months, required phosphate binder therapy, and had Medicare as their primary insurance.
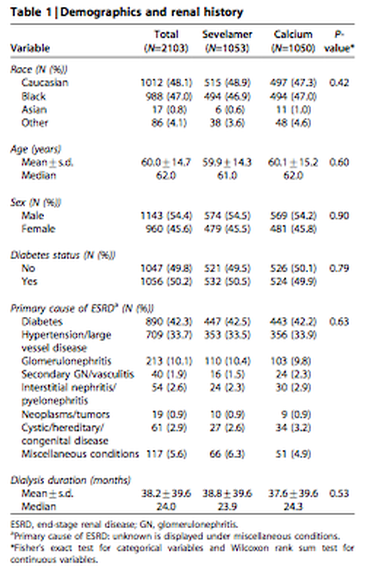
**The study demographics are shown in a chart below.
Intervention/Comparator
The DCOR (Dialysis Clinical Outcomes Revisited) trial compared all-cause mortality and cause-specific mortality (cardiovascular mortality, infection, and other causes) among hemodialysis patients treated with calcium-based phosphate binders (calcium carbonate and calcium acetate) and sevelamer.
Results
There was no difference in all-cause mortality between sevelamer group and calcium group (15/100 vs 16/100, p =0.40). No difference in mortality due to cardiovascular cause (8/100 sevelamer vs 8.6/100 calcium, p =0.53), mortality due to infection (2.6/100 sevelamer vs 2.4/100 calcium, p = 0.68) and mortality due to other causes (4.4/100 sevelamer vs 5.1/100 calcium, p = 0.33). There was no interaction between treatment and race, sex, diabetes status, cause of kidney disease or dialysis duration. Subgroup analysis showed in older patients (≥ 65 yo) in the sevelamer group, they had improved survival over the calcium group (18.2/100 vs 23.4/100, p = 0.02).
Of note the overall dropout rate was 47% in the sevelamer arm and 51% in the calcium-based binder arm. More patients in the sevelamer arm discontinued because of adverse events, which were mainly gastrointestinal. More patients in the calcium arm never took study medication or dropped out due to investigator decision.
Limitations
There were many limitations to this trial. There was a lack of blinding, a large drop-out rate and it was not an intention to treat model. The investigators determined the cause of death and decided when patients should discontinue therapy, which creates bias. There are missing baseline characteristics and assessment of compliance was inadequate. Finally concurrent therapies, such as vitamin D, were not monitored.
Bottom Line
There was no difference in all-cause mortality and cause-specific mortality between sevelamer HCl and calcium-based phosphate binders in dialysis patients. Sevelamer appears to be associated with improved survival in patients greater than or equal to 65 years old.
Effects of sevelamer and calcium-based phosphate binders on mortality in hemodialysis patients
Background
DCOR was an effectiveness trial that was designed to evaluate the treatment outcomes under usual care conditions in the hemodialysis population.
Trial Design
It was a multicenter, randomized, open-label effectiveness trial stratified by race (black and non-black), sex, diabetic status, and by age (<55 and >=55).
Population
This study included 2013 patients who were were >18 years, on dialysis for >3 months, required phosphate binder therapy, and had Medicare as their primary insurance.
**The study demographics are shown in a chart below.
Intervention/Comparator
The DCOR (Dialysis Clinical Outcomes Revisited) trial compared all-cause mortality and cause-specific mortality (cardiovascular mortality, infection, and other causes) among hemodialysis patients treated with calcium-based phosphate binders (calcium carbonate and calcium acetate) and sevelamer.
Results
There was no difference in all-cause mortality between sevelamer group and calcium group (15/100 vs 16/100, p =0.40). No difference in mortality due to cardiovascular cause (8/100 sevelamer vs 8.6/100 calcium, p =0.53), mortality due to infection (2.6/100 sevelamer vs 2.4/100 calcium, p = 0.68) and mortality due to other causes (4.4/100 sevelamer vs 5.1/100 calcium, p = 0.33). There was no interaction between treatment and race, sex, diabetes status, cause of kidney disease or dialysis duration. Subgroup analysis showed in older patients (≥ 65 yo) in the sevelamer group, they had improved survival over the calcium group (18.2/100 vs 23.4/100, p = 0.02).
Of note the overall dropout rate was 47% in the sevelamer arm and 51% in the calcium-based binder arm. More patients in the sevelamer arm discontinued because of adverse events, which were mainly gastrointestinal. More patients in the calcium arm never took study medication or dropped out due to investigator decision.
Limitations
There were many limitations to this trial. There was a lack of blinding, a large drop-out rate and it was not an intention to treat model. The investigators determined the cause of death and decided when patients should discontinue therapy, which creates bias. There are missing baseline characteristics and assessment of compliance was inadequate. Finally concurrent therapies, such as vitamin D, were not monitored.
Bottom Line
There was no difference in all-cause mortality and cause-specific mortality between sevelamer HCl and calcium-based phosphate binders in dialysis patients. Sevelamer appears to be associated with improved survival in patients greater than or equal to 65 years old.