Pathophysiology of Hyperphosphatemia (1)
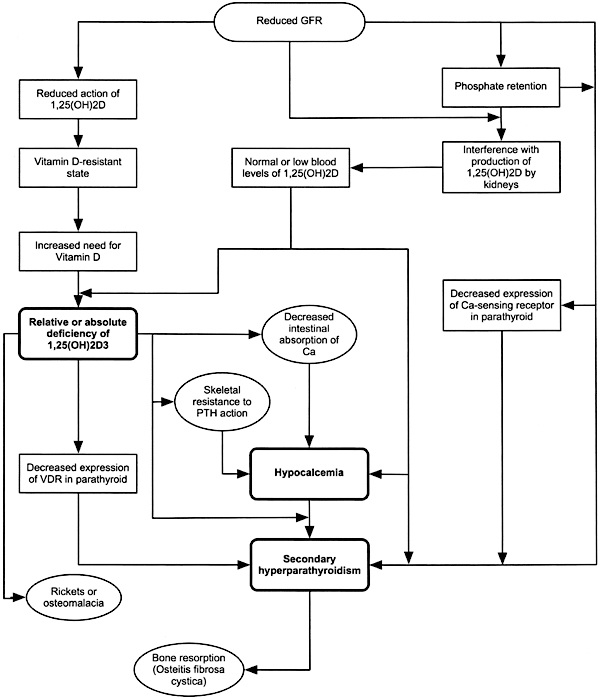
In patients with CKD decreased renal excretion of phosphate leads to phosphate retention. This interferes with the production of 1,25-dihydroxycholecalciferol (1,25 (OH) 2D3) by the kidneys.1 Vitamin D deficiency develops, which ultimately leads to decreased intestinal absorption of calcium and resultant hypocalcemia. Hyperphosphatemia and hypocalcemia both result in elevated blood levels of parathyroid hormone.1 As the kidneys continue to lose function, a decrease in vitamin D receptors and calcium-sensing receptors in the parathyroid glands occur. This results in increases in parathyroid hormone (PTH).1 Normally, PTH decreases the tubular reabsorption of phosphate by the kidney, increases urinary phosphate excretion, and consequently maintains serum phosphorus levels.1 Although phosphate retention occurs early in the course of CKD (Stage 2), hyperphosphatemia becomes evident in patients with marked loss of kidney function (Stage 4).1 Once kidney function decreases below 30 mL/min, the severely damaged kidneys cannot respond to further increments in PTH. The enhanced bone resorption, which occurs due to elevated levels of PTH, releases calcium and phosphorus from the skeleton into the extracellular fluid. The kidney cannot excrete this phosphorus and hence serum phosphorus concentration rises.1
In patients with CKD decreased renal excretion of phosphate leads to phosphate retention. This interferes with the production of 1,25-dihydroxycholecalciferol (1,25 (OH) 2D3) by the kidneys.1 Vitamin D deficiency develops, which ultimately leads to decreased intestinal absorption of calcium and resultant hypocalcemia. Hyperphosphatemia and hypocalcemia both result in elevated blood levels of parathyroid hormone.1 As the kidneys continue to lose function, a decrease in vitamin D receptors and calcium-sensing receptors in the parathyroid glands occur. This results in increases in parathyroid hormone (PTH).1 Normally, PTH decreases the tubular reabsorption of phosphate by the kidney, increases urinary phosphate excretion, and consequently maintains serum phosphorus levels.1 Although phosphate retention occurs early in the course of CKD (Stage 2), hyperphosphatemia becomes evident in patients with marked loss of kidney function (Stage 4).1 Once kidney function decreases below 30 mL/min, the severely damaged kidneys cannot respond to further increments in PTH. The enhanced bone resorption, which occurs due to elevated levels of PTH, releases calcium and phosphorus from the skeleton into the extracellular fluid. The kidney cannot excrete this phosphorus and hence serum phosphorus concentration rises.1
Phosphate Balance (2)
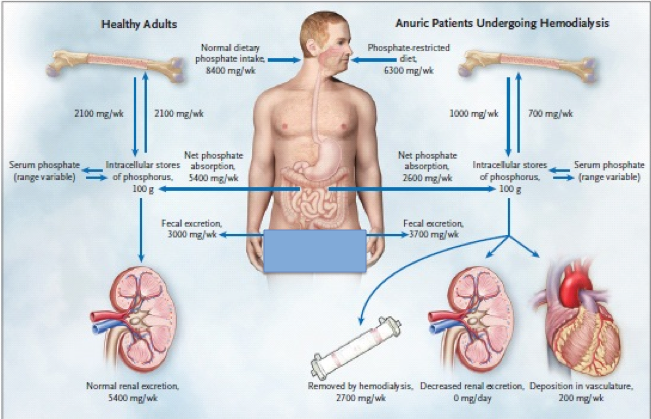
In individuals with working kidneys, phosphate intake is matched by phosphate excretion in feces and urine.2 The flux between the skeleton and extracellular phosphate is the same in both directions. In patients with chronic kidney failure, dietary restriction of phosphate is insufficient to compensate for the decrease in renal phosphate excretion, resulting in a positive phosphate balance.2 Bone is often resorbed more rapidly than it is formed because of abnormal bone remodelling. Together, these abnormalities may result in a predisposition of vascular calcification. The phosphate values shown are for illustrative purposes only as these values vary in individuals.
Hyperphosphatemia and Cardiovascular Disease (2)
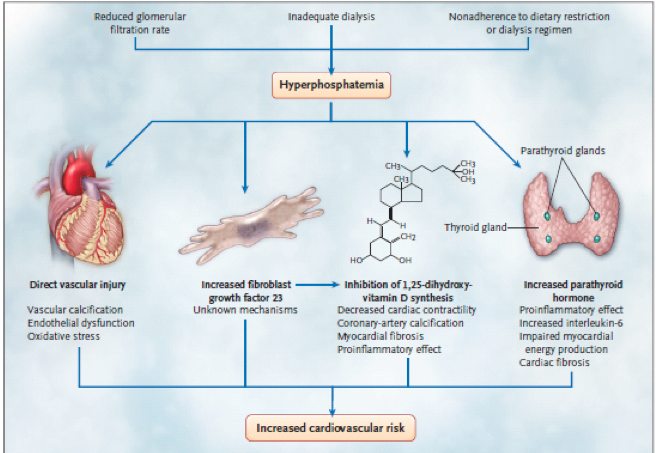
Elevated serum phosphate levels are linked to increased cardiovascular disease. It is unclear whether phosphate is a marker of poor outcome or plays a causal role.2 Multiple potential mechanims linking phosphate to cardiovascular disease exist. Hyperphosphatemia may directly affect vascular health by increasing reactive oxygen species, resulting in oxidative damage and endothelial dysfunction.2 Indirectly, hyperphosphatemia increases levels of fibroblast growth factor 23 and parathyroid hormone, which have both linked to cardiovascular disease.2 Increased phosphate levels have been associated with inhibition of 1,25 dihydroxyvitamin D synthesis, which is associated with vascular calcification and myocardial disease.1 These molecular mechanisms suggest that hyperphosphatemia may play a key role in progression of vascular calcification in CKD.
Elevated serum phosphate levels are linked to increased cardiovascular disease. It is unclear whether phosphate is a marker of poor outcome or plays a causal role.2 Multiple potential mechanims linking phosphate to cardiovascular disease exist. Hyperphosphatemia may directly affect vascular health by increasing reactive oxygen species, resulting in oxidative damage and endothelial dysfunction.2 Indirectly, hyperphosphatemia increases levels of fibroblast growth factor 23 and parathyroid hormone, which have both linked to cardiovascular disease.2 Increased phosphate levels have been associated with inhibition of 1,25 dihydroxyvitamin D synthesis, which is associated with vascular calcification and myocardial disease.1 These molecular mechanisms suggest that hyperphosphatemia may play a key role in progression of vascular calcification in CKD.
References
1. K/DOQI Clinical Practice Guidelines for Bone Metabolism and Disease in Chronic Kidney Disease. 2003. Web. 13 Jan 2013. <http://www.kidney.org/professionals/kdoqi/guidelines_bone/background.htm#fig1>
2. Tonelli M., Pannu N., & Manns B. (2010). Oral Phosphate Binders in Patients with Kidney Failure. NEJM, 362, 1312-24.
1. K/DOQI Clinical Practice Guidelines for Bone Metabolism and Disease in Chronic Kidney Disease. 2003. Web. 13 Jan 2013. <http://www.kidney.org/professionals/kdoqi/guidelines_bone/background.htm#fig1>
2. Tonelli M., Pannu N., & Manns B. (2010). Oral Phosphate Binders in Patients with Kidney Failure. NEJM, 362, 1312-24.