Chertow GM, Burke SK, Raggi P. Sevelamer attenuates the progression of coronary and aortic calcification in hemodialysis patients. Kidney Int 2002;62:245-252
Sevelamer attenuates the progression of coronary and aortic calcification in hemodialysis patients
Background
Cardiovascular disease is the leading cause of mortality in patients with end-stage renal disease. Mineral metabolism disorders (hyperparathyroidism and hyperphosphatemia) may contribute to cardiovascular calcification.
Trial Design
This was a randomized, open-label study comparing sevelamer hydrochloride and calcium salts (calcium acetate, calcium carbonate).
Population
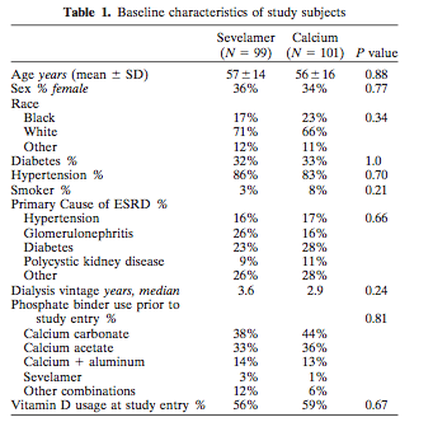
This study included 200 hemodialysis patients.
**The study demographics are shown in a chart below.
Intervention/Comparator
The study was designed to compare the effects of sevelamer hydrochloride against calcium-containing phosphate binders (calcium acetate and calcium carbonate) on serum phosphorus, calcium, calcium-phosphorus product and intact parathyroid hormone levels for one year.
Primary and Secondary Outcomes
The outcomes were the comparison of concentrations of serum phosphorus, calcium, intact parathyroid hormone (PTH), and calcification of the coronary arteries and thoracic aorta using a calcification score.
Results
Sevelamer and calcium provided equivalent control of serum phosphorus (end-of-study values 1.65 ± 0.39 and 1.65 ± 0.45 mmol/L, respectively, P = 0.33). Serum calcium concentration was significantly higher in the calcium-treated group (P = 0.002), and hypercalcemia was more common (16% vs. 5% with sevelamer, P = 0.04).
Limitations
This was a non-blinded study that did not control for lipid levels, vitamin D dose or dialysate calcium levels. The calcium products being compared were not the same (In US calcium acetate was used and in Europe calcium carbonate used).
Bottom Line
Sevelamer is less likely to cause hypercalcemia, low levels of PTH, and progressive coronary and aortic calcification in hemodialysis patients (duration ~ 3 years).
Both agents controlled serum phosphorous equally.
Sevelamer attenuates the progression of coronary and aortic calcification in hemodialysis patients
Background
Cardiovascular disease is the leading cause of mortality in patients with end-stage renal disease. Mineral metabolism disorders (hyperparathyroidism and hyperphosphatemia) may contribute to cardiovascular calcification.
Trial Design
This was a randomized, open-label study comparing sevelamer hydrochloride and calcium salts (calcium acetate, calcium carbonate).
Population
This study included 200 hemodialysis patients.
**The study demographics are shown in a chart below.
Intervention/Comparator
The study was designed to compare the effects of sevelamer hydrochloride against calcium-containing phosphate binders (calcium acetate and calcium carbonate) on serum phosphorus, calcium, calcium-phosphorus product and intact parathyroid hormone levels for one year.
Primary and Secondary Outcomes
The outcomes were the comparison of concentrations of serum phosphorus, calcium, intact parathyroid hormone (PTH), and calcification of the coronary arteries and thoracic aorta using a calcification score.
Results
Sevelamer and calcium provided equivalent control of serum phosphorus (end-of-study values 1.65 ± 0.39 and 1.65 ± 0.45 mmol/L, respectively, P = 0.33). Serum calcium concentration was significantly higher in the calcium-treated group (P = 0.002), and hypercalcemia was more common (16% vs. 5% with sevelamer, P = 0.04).
Limitations
This was a non-blinded study that did not control for lipid levels, vitamin D dose or dialysate calcium levels. The calcium products being compared were not the same (In US calcium acetate was used and in Europe calcium carbonate used).
Bottom Line
Sevelamer is less likely to cause hypercalcemia, low levels of PTH, and progressive coronary and aortic calcification in hemodialysis patients (duration ~ 3 years).
Both agents controlled serum phosphorous equally.